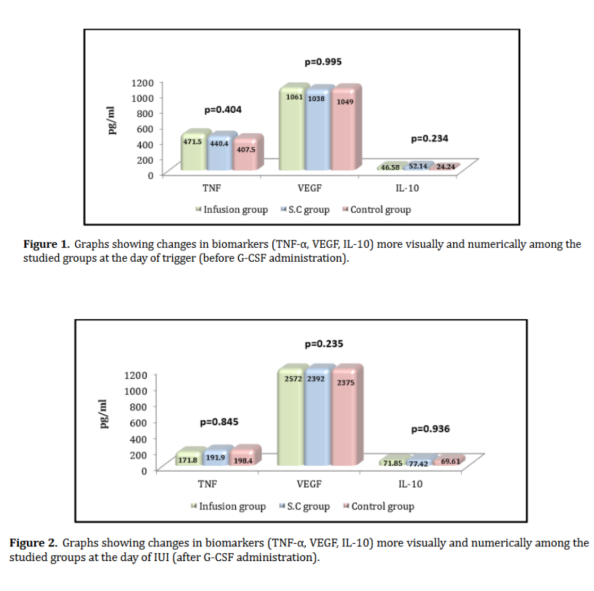
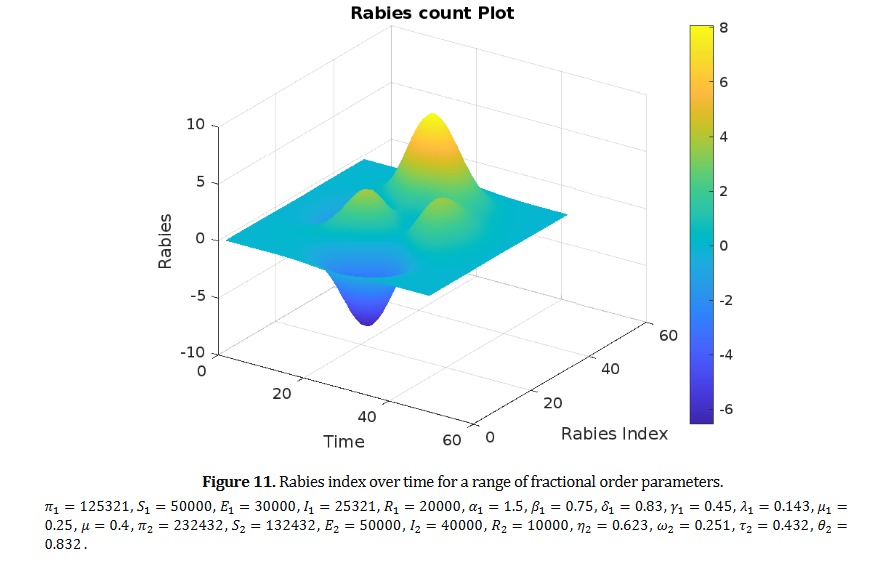
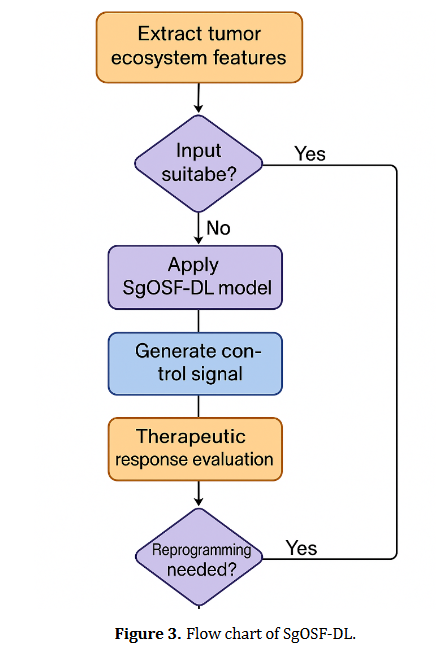
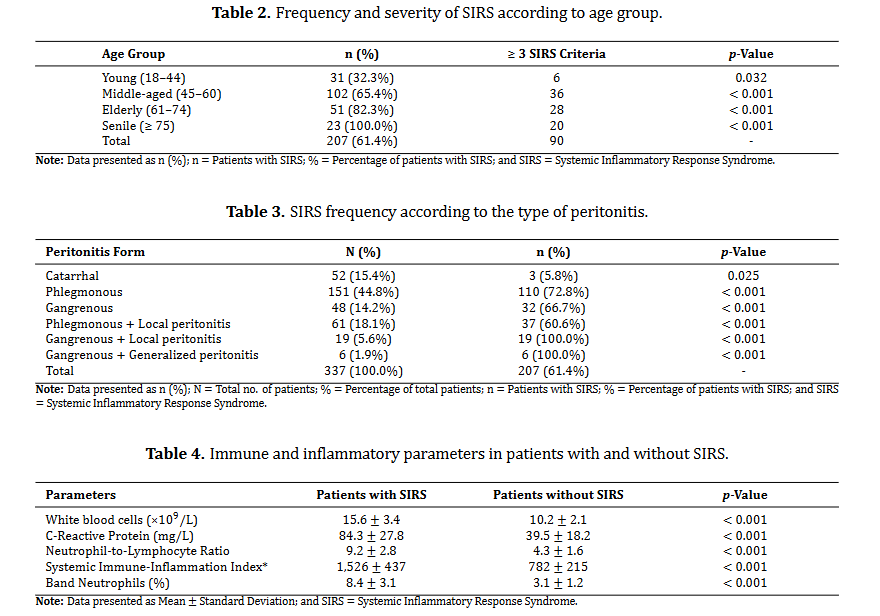
Genital endometriosis is a chronic inflammatory condition characterized by immune dysfunction involving both innate and adaptive immunity. Disrupted immune responses, particularly those involving macrophages, dendritic cells, natural killer cells, regulatory T cells, T helper 17 cells, and B-cell-driven autoimmunity, create an environment conducive to ectopic endometrial tissue survival. The cytokine milieu, marked by elevated levels of interleukin-6, tumor necrosis factor-alpha, and interleukin-10, facilitates the interaction between inflammation and immune tolerance, thereby driving disease progression. Current hormonal and surgical treatments offer only temporary or partial symptom relief and fail to address the underlying immunopathogenic mechanisms of the disease. Their limitations, including high recurrence rates, systemic side effects, and insufficient fertility restoration, highlight the need for novel interventions that target specific pathways. Recent advances in immunology and microbiome research have led to promising therapeutic strategies, such as microbiome modulation, precision medicine based on immune phenotyping, and integrative care models that provide personalized and comprehensive treatment. Future research should focus on advancing immune and microbial profiling to guide targeted therapies, validating immunomodulatory approaches, and integrating these methods into clinical practices. By bridging basic research with clinical applications, the field is poised to shift from managing symptoms to altering the disease trajectory, ultimately improving outcomes for women affected by genital endometriosis.
 626 (Views)
626 (Views)  395 (Downloads)
395 (Downloads)