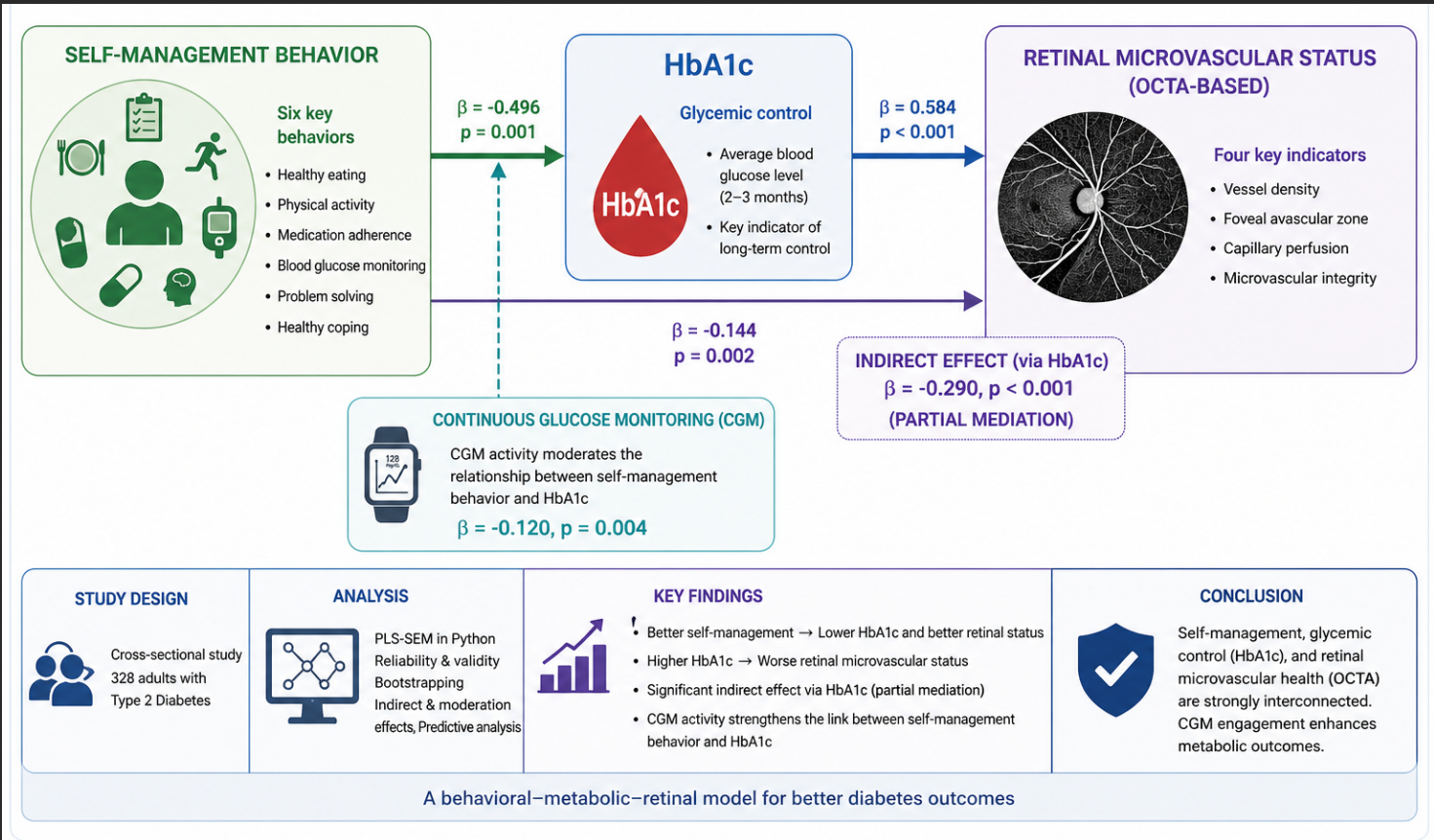
The study evaluates the relationship with diabetes self-management behavior and retinal microvascular status and hypothesizes whether HbA1c has an indirect statistically significant association between diabetes self-management behavior and retinal microvascular status in individuals with type 2 diabetes. The second view is to determine whether the involvement of continuous glucose monitoring is a moderator of the relationship between the self-management behavior and the HbA1c. An empirical cross-sectional study involved 328 adults with type 2 diabetes. The model combined six indicators of self-management, three of the HbA1c indicators, four indicators of the Optical Coherence Tomography Angiography (OCTA)-based retinal microvascular indicators, and four indicators of the continuous glucose monitor activity. A Partial Least Squares Structural Equation Modeling (PLS-SEM) model was developed in Python and evaluated based on reliability, convergent and discriminant validity, bootstrapped path estimation, indirect effect test, moderation analysis, and predictive analysis. Better self-management behavior is related to lower HbA1c (β = −0.496, p = 0.001) and more desirable retinal microvascular situation (β = −0.144, p = 0.002). Worse retinal microvascular status was linked with higher levels of HbA1c (β = 0.584, p < 0.001). The self-management behavior indirectly related to retinal microvascular status via HbA1c was significant (β = −0.290, p < 0.001), and this could be attributed to partial mediation. The constant glucose monitoring involvement had a significant moderate effect on the association between self-management behavior and HbA1c (β = −0.120, p = 0.004). The results substantiate a composite behavioral, metabolic, and retinal imaging model with self-management behavior, HbA1c, and OCTA-generated retinal radiations being strongly correlated.
 129 (Views)
129 (Views)  68 (Downloads)
68 (Downloads)