Review Article ID: 1670
by Hayat Alshahrani,
Areej Alshammari,
Shoug Alshammari,
Zainab Alshaikh,
Lujain Alahamdi,
Wajan Alqathanin,
Wafaa Alnujaimi,
Fatimah Alfaqih,
Shahad Bandar,
Leen Al Malawi,
Lama Altuwijri,
Raghida Almuzaini,
Yazeed A. Alhelali,
Sahar Alward,
Kholoud Alshahrani
Volume 15 Issue 4 (2025). 52-64. DOI:
https://doi.org/10.54963/entu.v15i4.1670
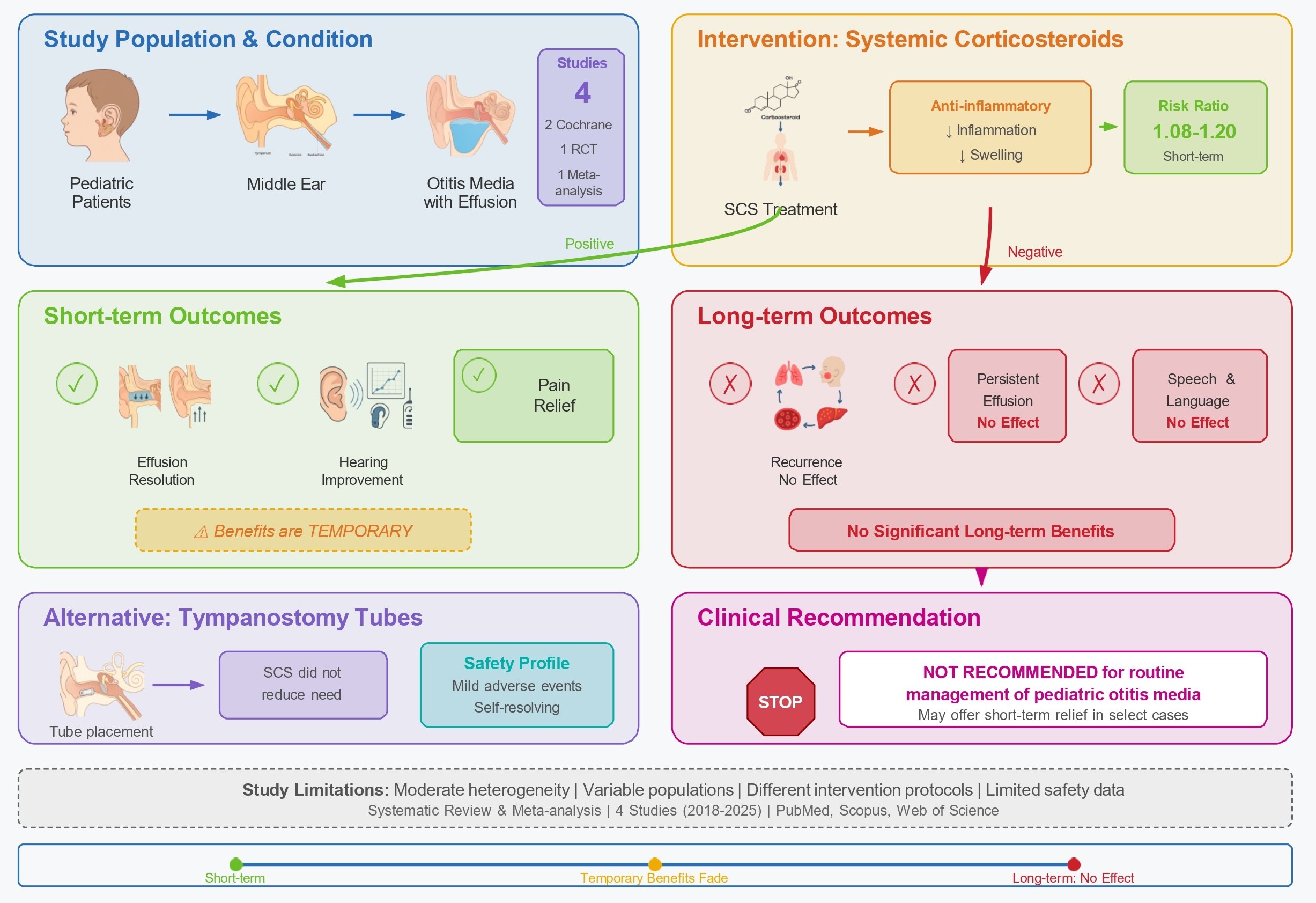
Otitis media is a common childhood condition that affects public health because of its frequency, recurrence, and potential long-term consequences. This systematic review and meta-analysis assessed the efficacy and safety of systemic corticosteroids (SCS) in children with acute otitis media and otitis media with an effusion. A comprehensive search of PubMed, Scopus, and Web of Science identified relevant studies published between January 2018 and August 2025. The primary outcomes were effusion resolution, hearing loss, and otitis media recurrence. Secondary outcomes included pain relief, tympanostomy tube placement, speech and language development, quality of life, and adverse events. Four studies, including two Cochrane reviews, one randomized controlled trial, and one meta-analysis, were analyzed. The results showed that SCS provided short-term improvements in effusion clearance and hearing, with risk ratios from 1.08 to 1.20. However, these benefits were temporary, with no significant long-term effects on recurrence rates, persistent effusion, or developmental outcomes. Safety data were limited, with most adverse events being mild and self-resolving. The studies had moderate heterogeneity due to differences in population characteristics, intervention protocols, and outcome definitions. Results suggest that SCS may offer short-term symptom relief in certain cases but cannot be recommended for routine management of pediatric otitis media. Future research should prioritize large-scale multicenter trials with standardized outcomes, extended follow-up periods, and thorough safety assessments to identify the subgroups that might benefit the most from SCS treatment.

 381 (Views)
381 (Views)  144 (Downloads)
144 (Downloads)