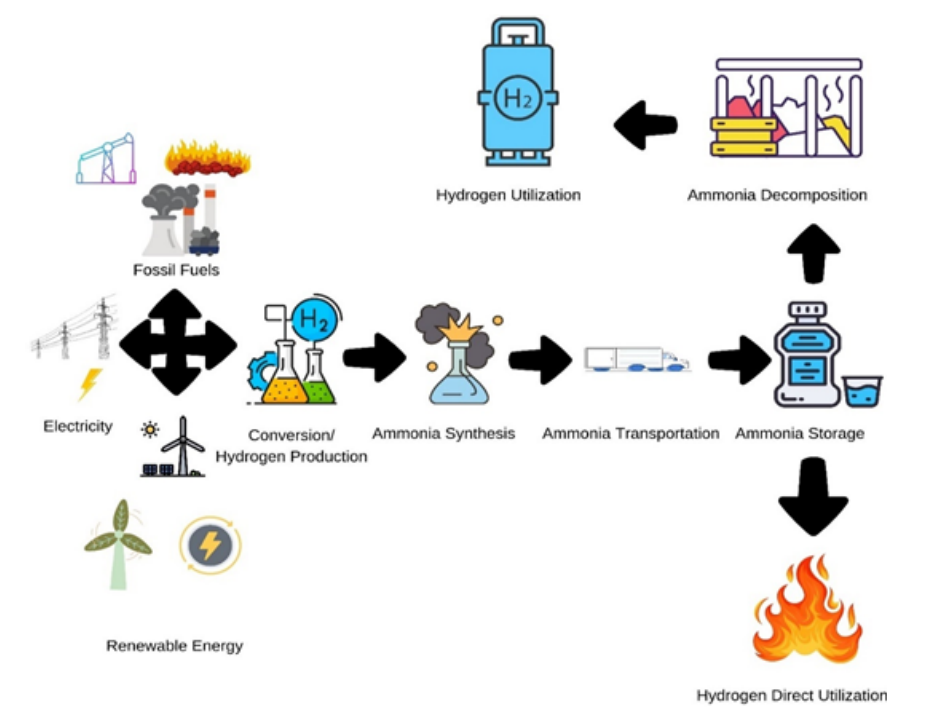
The simple storage of ammonia, combined with the tendency to liberate hydrogen without carbon dioxide emissions, has made ammonia breakdown popular among the research community in recent years. It has evolved as a promising method for hydrogen (H₂) production. The discussion highlights the critical role of catalyst composition, support materials, and promoters in enhancing activity, stability, and scalability. By comparing the advantages and limitations of each method, this work provides a roadmap for future research aimed at optimising NH₃-to-H₂ conversion for sustainable energy applications. This review article has discussed the current advances in ammonia breakdown technology for hydrogen generation, focusing on new materials and mechanical designs for catalysis especially emphasising thermocatalytic, photocatalytic, and electrocatalytic methods. Importance is given to exploring the recent developments of efficient and cost-effective catalysts, including monometallic (e.g., Ru, Ni) and bimetallic systems (e.g., Ru-Ni, Ni-Co), as well as metal hydrides. The Challenges, like high reaction temperatures, slow kinetics, and catalyst deactivation, are explored, and prospective solutions, such as low-temperature oxidative cleavage and plasma-assisted methods. The review also explores the mechanistic insights into NH₃ decomposition pathways and the synergistic effects of bimetallic catalysts. Moreover, it would help to update the knowledge about the catalytic reaction processes and emphasise the benefits and drawbacks of each strategy. Furthermore, the significance of discovering a cost-effective metal catalyst with better efficiency and higher reliability is also debated. This article may serve as a fundamental resource to scale up information about the catalytic production of hydrogen from ammonia.

 185 (Views)
185 (Views)  125 (Downloads)
125 (Downloads)